FreeDM2: Real-time CGM for type 2 diabetes on basal insulin.

A UK randomised trial in the largest insulin-treated type 2 cohort that NICE guidelines NG28 doesn't currently fund.
FreeDM2: Real-time CGM for type 2 diabetes on basal insulin shows a gap in NICE eligibility and evidence.
In UK primary care, continuous glucose monitoring is now standard for type 1 diabetes. It is also funded for certain type 2 patients who use multiple daily injections. Patients on basal insulin alone make up the largest group of insulin-treated type 2 diabetes patients in primary care. Right now, they are not included in that eligibility envelope. The FreeDM2 trial, published in The Lancet Diabetes and Endocrinology on April 23, 2026, is the first UK randomised controlled trial. It tests whether real-time CGM benefits this group. It was also presented at the Diabetes UK Professional Conference in Liverpool that week.¹
Around 18 to 30% of people on basal insulin reach their HbA1c targets in real-world data. National guidance has been careful about expanding CGM use beyond multiple daily injections. This caution comes from limited trial evidence in basal-insulin-only groups. FreeDM2 was designed to address that gap.
The trial randomised 303 adults across 24 UK clinical sites. Eligible participants had type 2 diabetes and were on basal insulin. They could also be using other glucose-lowering therapies, like SGLT2 inhibitors or GLP-1 receptor agonists. Their HbA1c was above their personal target at the start. Participants were assigned to either real-time CGM using the Abbott FreeStyle Libre system or to traditional self-monitoring of blood glucose by fingerstick. The trial ran in two phases. During the first 16 weeks, participants managed their diabetes on their own. They used their assigned monitoring methods and didn’t change medications based on a protocol. In the subsequent 16 weeks, treatment intensification was supported by clinicians as clinically indicated. The primary outcome was change in HbA1c at 16 weeks.
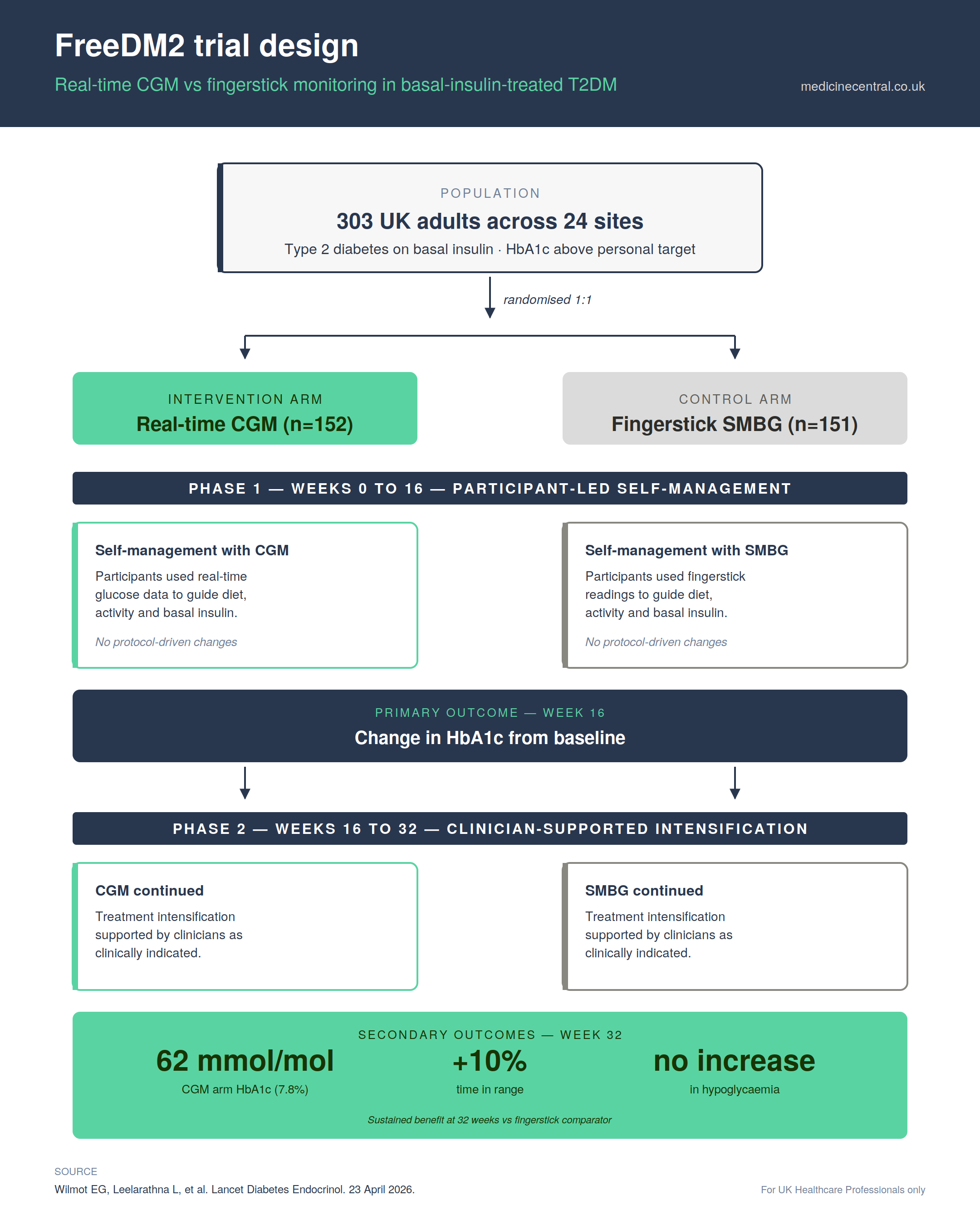
At 16 weeks, HbA1c fell by 9 mmol/mol (0.8%) in the CGM arm and by 2 mmol/mol (0.1%) in the fingerstick arm. A difference of 7 mmol/mol was both statistically significant and clinically important. This level of reduction lowers the risk of long-term microvascular complications. This difference showed up during the self-management phase. There were no major differences in insulin doses between the groups. Participants used CGM-derived insights to make adjustments to diet, activity and basal insulin themselves.
The benefit was sustained at 32 weeks. Mean HbA1c was 62 mmol/mol (7.8%) in the CGM arm versus 67 mmol/mol (8.3%) in the fingerstick arm. Time in range (3.9 to 10.0 mmol/L) was approximately 10% higher in the CGM group, equivalent to around 2.5 additional hours per day in target. There was no increase in hypoglycaemia in the CGM arm. Participants already taking SGLT2 inhibitors or GLP-1 receptor agonists at baseline still showed meaningful HbA1c improvement with CGM. The benefit was not contingent on adding new medication classes, which speaks to a behavioural mechanism rather than a pharmacological one.
Caveats sharpen interpretation:
The trial was open-label, which is unavoidable for monitoring interventions. Participants and clinicians knew their assignments. This may have affected behavior and clinical decisions in the second phase.
Formal cost-effectiveness analysis in the UK setting has not yet been published. Diabetes UK has clearly stated that this analysis must happen first. Only then can broader access on NHS terms be considered.
Current NICE NG28 criteria for CGM in type 2 diabetes focus on:
Patients on multiple daily injection regimens
Those with disabling hypoglycemia
Individuals who cannot self-monitor by fingerstick
Patients on basal insulin alone, even where HbA1c is above target, are not generally eligible. FreeDM2 targets this overlooked group and offers benefits similar to many drugs. This does not change NG28 today. Guidelines do not move on a single trial, however well designed. What it does change is the conversation. Patients on basal insulin can now ask prescribers about CGM. There are UK randomised data available, but keep in mind that funding and access haven’t caught up yet. This trial is key for practices auditing their basal-insulin type 2 cohorts before the next NG28 review. Committees will definitely look at this evidence.
A formal cost-effectiveness analysis is the next step. NICE hasn't announced a review of NG28 CGM eligibility yet. However, the FreeDM2 data and the Italian observational evidence from ATTD earlier this year will be part of any future re-evaluation (NCT06268808).
Local ICBs may also begin to reflect this data in their diabetes formularies and shared care arrangements ahead of any national change.
For background on related kidney and cardiovascular endpoints in this same patient cohort, see our earlier coverage of SGLT2i vs GLP-1 RA kidney protection and the NICE NG28 update.
References
Wilmot EG, Leelarathna L, et al. Real-time continuous glucose monitoring versus self-monitoring of blood glucose in adults with type 2 diabetes treated with basal insulin (FreeDM2): a randomised controlled trial. The Lancet Diabetes and Endocrinology. Published 23 April 2026.
Medicine Central is a clinical evidence review for UK primary care clinicians. Content reflects evidence current at time of publication and should be read alongside local formulary and clinical guidance. Guest contributors retain responsibility for the accuracy and originality of their work. Views expressed are the author's own and do not necessarily reflect those of Medicine Central. For healthcare professionals only.
Enjoyed this article?
- ✓Unlimited access to clinical evidence articles
- ✓Weekly newsletter curated for UK primary care
- ✓Free for registered UK healthcare professionals
